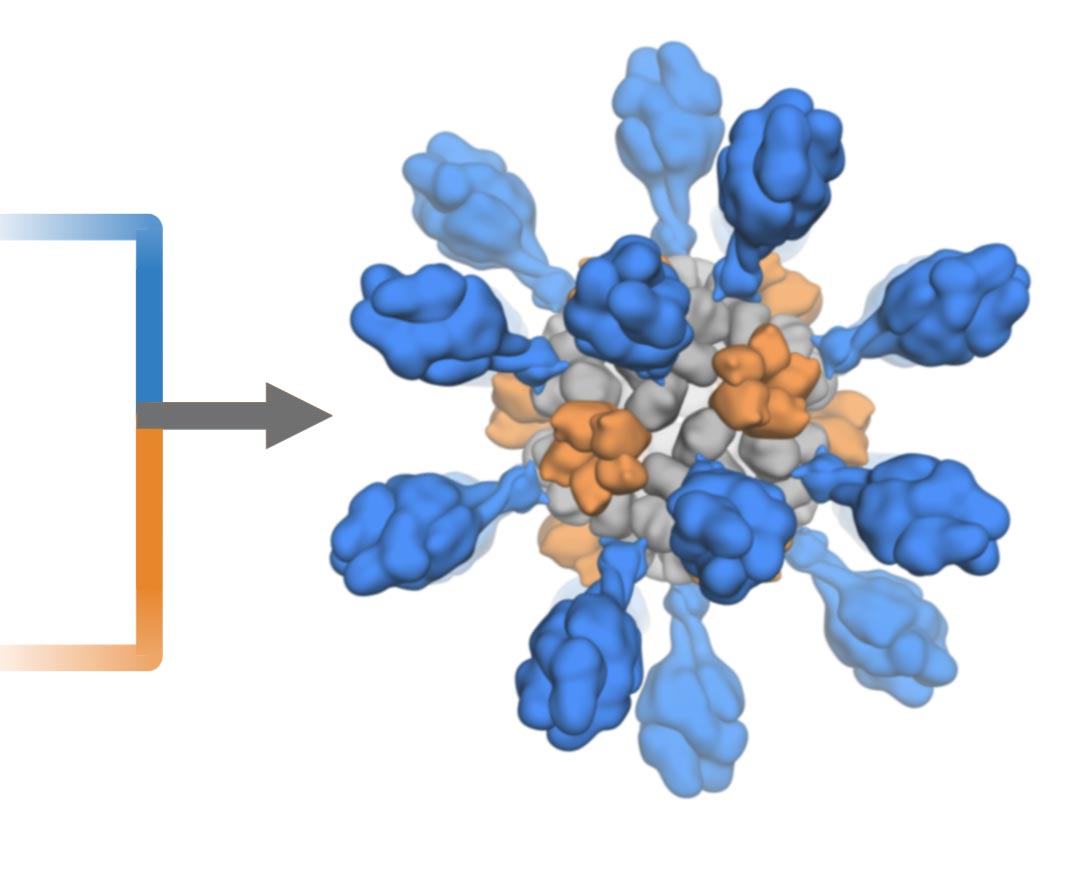
Artist’s impression of an ultrapotent COVID-19 vaccine candidate in which 60 pieces of a coronavirus protein (red) decorate nanoparticles (blue and white). The vaccine candidate was designed using methods developed at the UW Medicine Institute for Protein Design. The vaccine’s molecular structure mimics that of a virus, which may be responsible for its improved ability to trigger an immune response. Photo credit: Ian Haydon / UW Medicine Institute for Protein Design
Preclinical data published in Cell shows that the nanoparticle vaccine elicits extremely high levels of protective antibodies in animal models.
An innovative nanoparticle vaccine candidate for the pandemic coronavirus produces virus-neutralizing antibodies in mice in concentrations ten times higher than in people who have recovered from them COVID-19 Infections. Designed by scientists of the University of Washington The Seattle School of Medicine vaccine candidate has been transferred to two companies for clinical development.
Compared to vaccination with the soluble SARS-CoV-2 Spike protein, on which many leading COVID-19 vaccine candidates are based, the new nanoparticle vaccine produced ten times neutralizing antibodies in mice, even at a six times lower dose. The data also show a strong B-cell response after immunization, which can be critical for immune memory and a lasting vaccine effect. When administered to a single non-human primate, the nanoparticle vaccine produced neutralizing antibodies that targeted several different sites on the spike protein. Researchers say this could provide protection from mutant strains of the virus should they arise. The spike protein is part of the coronavirus infectivity machinery.
The results were published on October 30, 2020 in cell. The main authors of this work are Alexandra Walls, a scientist in the laboratory of David Veesler, an Associate Professor of Biochemistry at UW; and Brooke Fiala, a scientist in the laboratory of Neil King, UW Assistant Professor of Biochemistry.
The vaccine candidate was developed using structure-based vaccine design techniques invented at UW Medicine. It is a self-assembling protein nanoparticle that displays 60 copies of the receptor binding domain of the SARS-CoV-2 spike protein in a highly immunogenic array. The vaccine’s molecular structure mimics that of a virus, which may be responsible for its improved ability to trigger an immune response.
“We hope that our nanoparticle platform can help fight this pandemic that is causing so much damage to our world,” said King, inventor of computational vaccine design technology at the Institute for Protein Design. “The efficacy, stability and manufacturability of this vaccine candidate sets it apart from many others studied.”
Hundreds of vaccine candidates for COVID-19 are being developed worldwide. Many require large doses, complex manufacturing, and shipping and cold chain storage. An ultrapotent vaccine that is safe, effective at low doses, easy to make and stable outside of a freezer could enable vaccination against COVID-19 on a global scale.
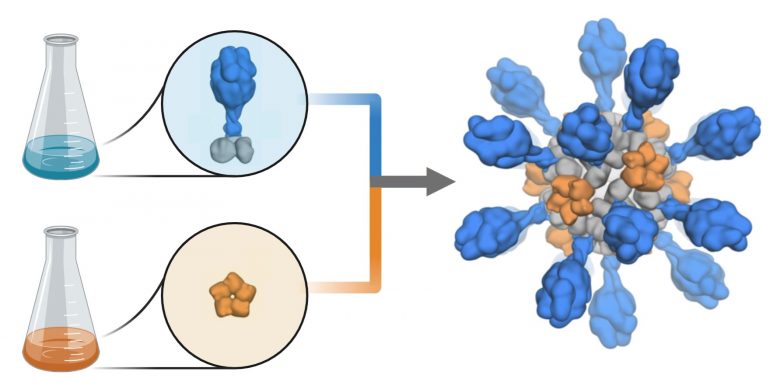
The production scheme shows how coronavirus proteins are added to a computerized nanoparticle platform to create a vaccine candidate against COVID-19. The vaccine candidate was designed and tested in animal models by researchers at the University of Washington School of Medicine. Photo credit: Ian Haydon / UW Medicine Institute for Protein Design
“I am delighted that our studies of antibody responses to coronaviruses have led to the design of this promising vaccine candidate,” said Veesler, who led the concept of a multivalent, receptor-binding, domain-based vaccine.
The lead vaccine candidate from this report will not be licensed exclusively and royalty-free from the University of Washington during the pandemic. A licensee, Icosavax, a Seattle-based biotechnology company co-founded by King in 2019, is currently advancing studies to support regulatory filings and has initiated the U.S. Food and Drug Administration’s Good Manufacturing Practice. To accelerate Icosavax’s progress in the clinic, Amgen has agreed to manufacture a key intermediate for these initial clinical trials. Another licensee, SK Bioscience from South Korea, is promoting its own studies to support clinical and further development.
Reference: “Induction of strong neutralizing antibody responses through designed protein nanoparticle vaccines for SARS-CoV-2, Alexandra C. Walls, Brooke Fiala, Alexandra Schäfer, Samuel Wrenn, Minh N. Pham, Michael Murphy, Longping V. Tse, Laila Shehata, Megan A. O’Connor, Chengbo Chen, Mary Jane Navarro, Marcos C. Miranda, Deleah Pettie, Rashmi Ravichandran, John C. Kraft, Cassandra Ogohara, Anne Palser, Sara Kreide, E-Chiang Lee, Kathryn Guerriero, Elizabeth Kepl, Cameron M. Chow, Claire Sydeman, Edgar A. Hodge, Brieann Brown, Jim T. Fuller, Kenneth H. Dinnon III, Lisa E. Gralinski, Sarah R. Leist, Kendra L. Gully, Thomas B. Lewis, Miklos Guttman, Helen Y. Chu, Kelly K. Lee, Deborah H. Fuller, Ralph S. Baric, Paul Kellam, Lauren Carter, Marion Pepper, Timothy P. Sheahan, David Veesler and Neil P. King, accepted October 26, 2020, cell.
DOI: 10.1016 / j.cell.2020.10.043
Research reported in Cell was funded by the National Institute for Allergies and Infectious Diseases (DP1AI158186, HHSN272201700059C, 3U01AI42001-02S1), the National Institute of General Medical Sciences (R01GM120553, R01GM099989), Bill & Melinda Gates Reduction Agency (OPP211511), Defense Agency 526 (HDTRA1-18-1-0001), Pew Biomedical Scholars Award, Burroughs Wellcome Fund Investigator Award, Fast Grants, Animal Model Contract HHSN272201700036I-75N93020F00001, University of Washington Proteomics Resource (UWPR95794), Relief Coronavirus, North Carolina Fund and gifts from Jodi Green and Mike Halperin as well as from The Audacious Project.